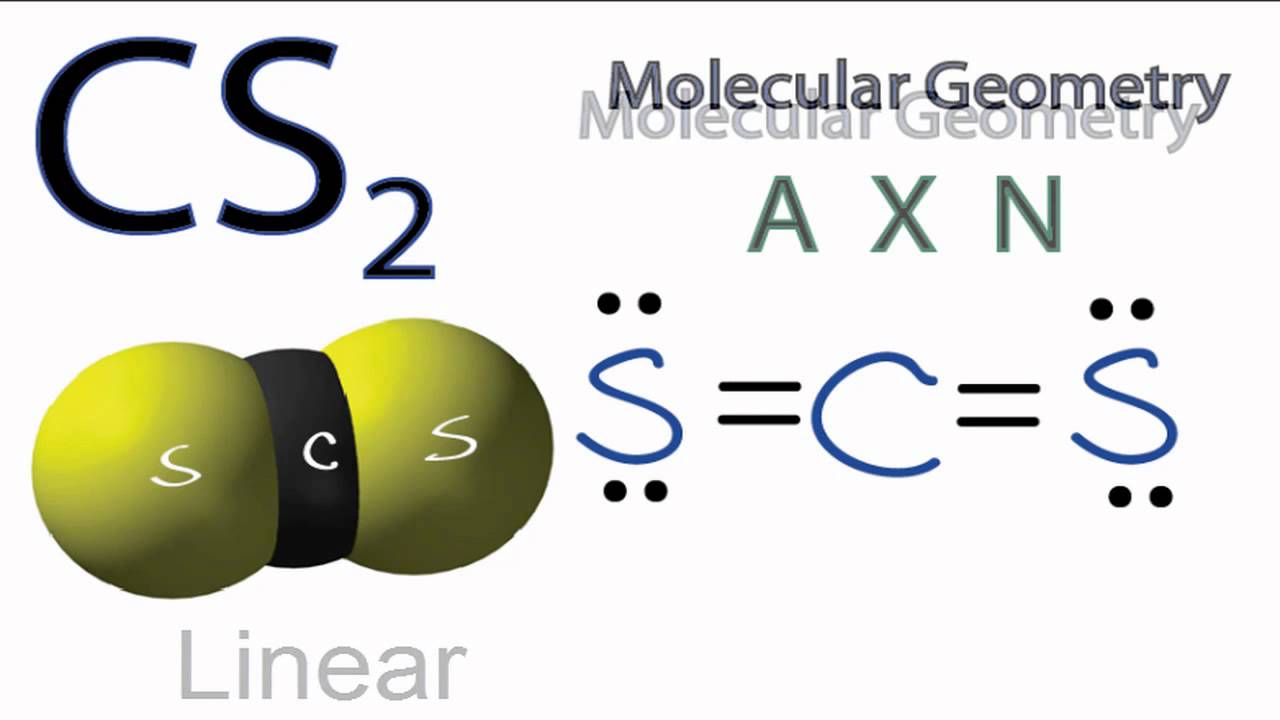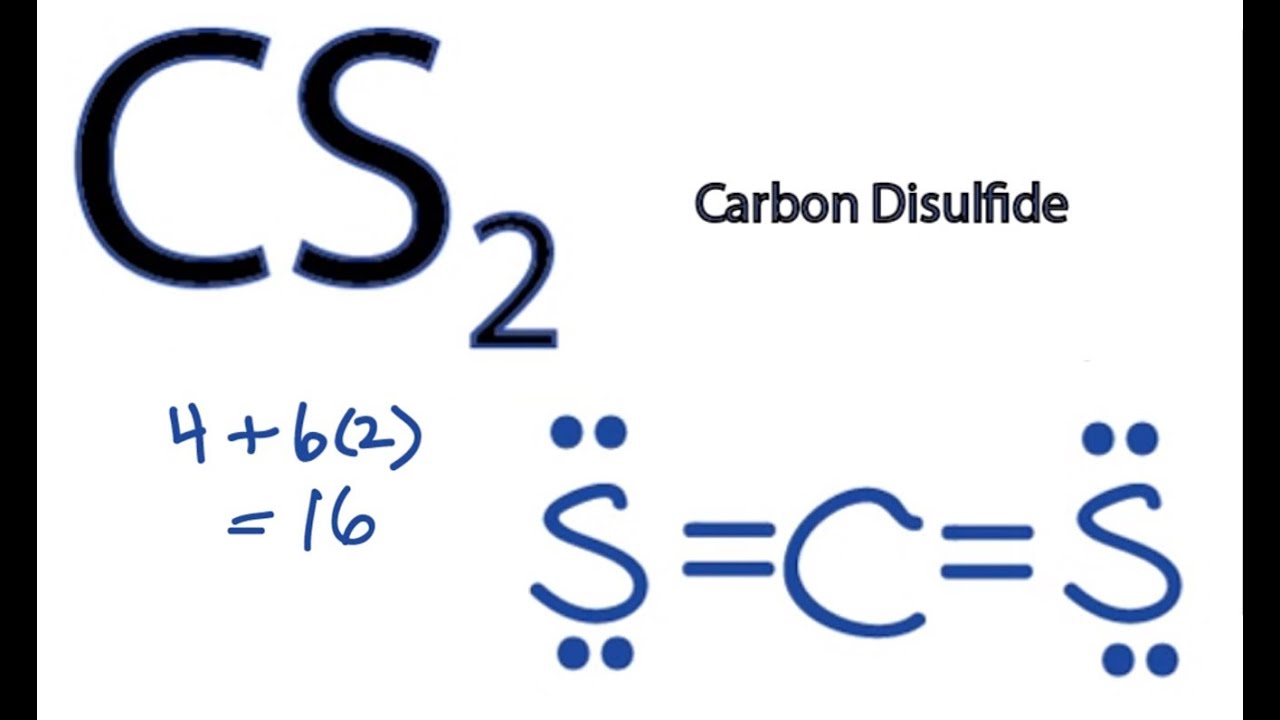What Is The Molecular Geometry Of Cs2
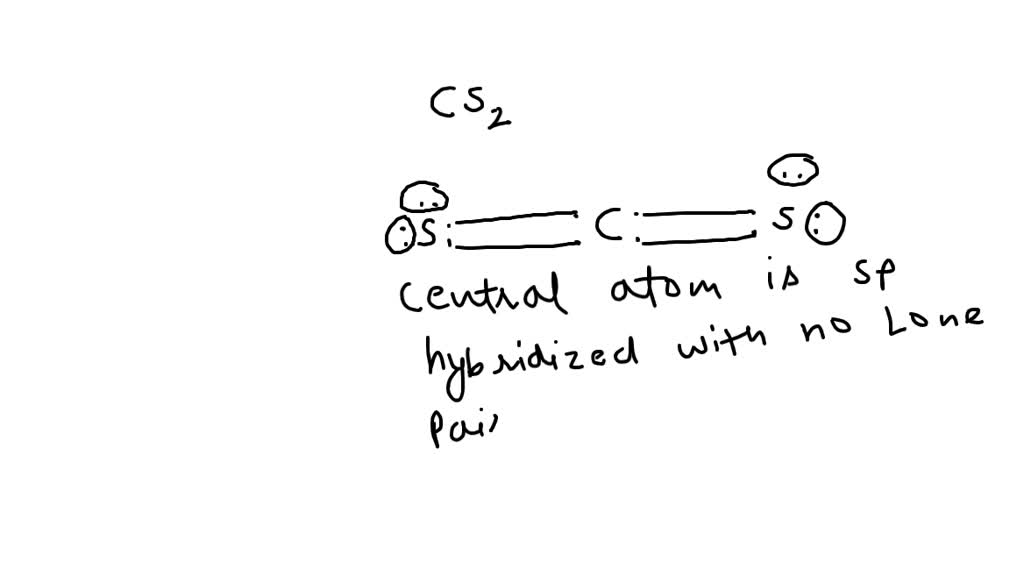
What Is The Molecular Geometry Of Cs2 - As the hybridization of cs2 is sp hybridization, the carbon atom is in center bonding with two sulfur atoms. Carbon disulfide, cs2, will have a total of 16 valence electrons, 4 from the carbon atom and 6 from each of the two sulfur atoms.
Carbon disulfide, cs2, will have a total of 16 valence electrons, 4 from the carbon atom and 6 from each of the two sulfur atoms. As the hybridization of cs2 is sp hybridization, the carbon atom is in center bonding with two sulfur atoms.
As the hybridization of cs2 is sp hybridization, the carbon atom is in center bonding with two sulfur atoms. Carbon disulfide, cs2, will have a total of 16 valence electrons, 4 from the carbon atom and 6 from each of the two sulfur atoms.
CS2 Molecular Geometry / Shape and Bond Angles YouTube
Carbon disulfide, cs2, will have a total of 16 valence electrons, 4 from the carbon atom and 6 from each of the two sulfur atoms. As the hybridization of cs2 is sp hybridization, the carbon atom is in center bonding with two sulfur atoms.
CS2 Lewis Structure How to Draw the Lewis Structure for CS2 YouTube
Carbon disulfide, cs2, will have a total of 16 valence electrons, 4 from the carbon atom and 6 from each of the two sulfur atoms. As the hybridization of cs2 is sp hybridization, the carbon atom is in center bonding with two sulfur atoms.
CS2 Molecular Geometry,Shape and Bond Angles (Carbon Disulfide) YouTube
Carbon disulfide, cs2, will have a total of 16 valence electrons, 4 from the carbon atom and 6 from each of the two sulfur atoms. As the hybridization of cs2 is sp hybridization, the carbon atom is in center bonding with two sulfur atoms.
10+ Molecular Geometry Of Cs2 Full Metry
Carbon disulfide, cs2, will have a total of 16 valence electrons, 4 from the carbon atom and 6 from each of the two sulfur atoms. As the hybridization of cs2 is sp hybridization, the carbon atom is in center bonding with two sulfur atoms.
CS2 Lewis Structure, Hybridization, Polarity and Molecular Shape
Carbon disulfide, cs2, will have a total of 16 valence electrons, 4 from the carbon atom and 6 from each of the two sulfur atoms. As the hybridization of cs2 is sp hybridization, the carbon atom is in center bonding with two sulfur atoms.
(Get Answer) The Molecular Geometry Of The CS2 Molecule Is O A. Bent
As the hybridization of cs2 is sp hybridization, the carbon atom is in center bonding with two sulfur atoms. Carbon disulfide, cs2, will have a total of 16 valence electrons, 4 from the carbon atom and 6 from each of the two sulfur atoms.
SOLVED What is the molecular geometry of CS2?
As the hybridization of cs2 is sp hybridization, the carbon atom is in center bonding with two sulfur atoms. Carbon disulfide, cs2, will have a total of 16 valence electrons, 4 from the carbon atom and 6 from each of the two sulfur atoms.
H2CO Molecular Geometry, Bond Angles & Electron Geometry (Formaldehyde
As the hybridization of cs2 is sp hybridization, the carbon atom is in center bonding with two sulfur atoms. Carbon disulfide, cs2, will have a total of 16 valence electrons, 4 from the carbon atom and 6 from each of the two sulfur atoms.
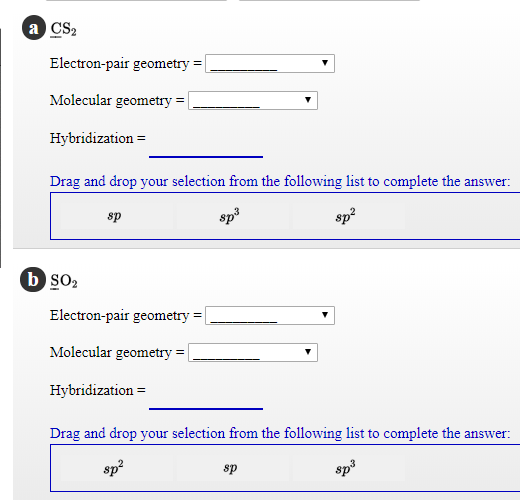
Solved CS2 Electronpair geometry Molecular geometry
Carbon disulfide, cs2, will have a total of 16 valence electrons, 4 from the carbon atom and 6 from each of the two sulfur atoms. As the hybridization of cs2 is sp hybridization, the carbon atom is in center bonding with two sulfur atoms.
SOLVED What is the molecular geometry (molecular structure) of CS2
Carbon disulfide, cs2, will have a total of 16 valence electrons, 4 from the carbon atom and 6 from each of the two sulfur atoms. As the hybridization of cs2 is sp hybridization, the carbon atom is in center bonding with two sulfur atoms.
Carbon Disulfide, Cs2, Will Have A Total Of 16 Valence Electrons, 4 From The Carbon Atom And 6 From Each Of The Two Sulfur Atoms.
As the hybridization of cs2 is sp hybridization, the carbon atom is in center bonding with two sulfur atoms.