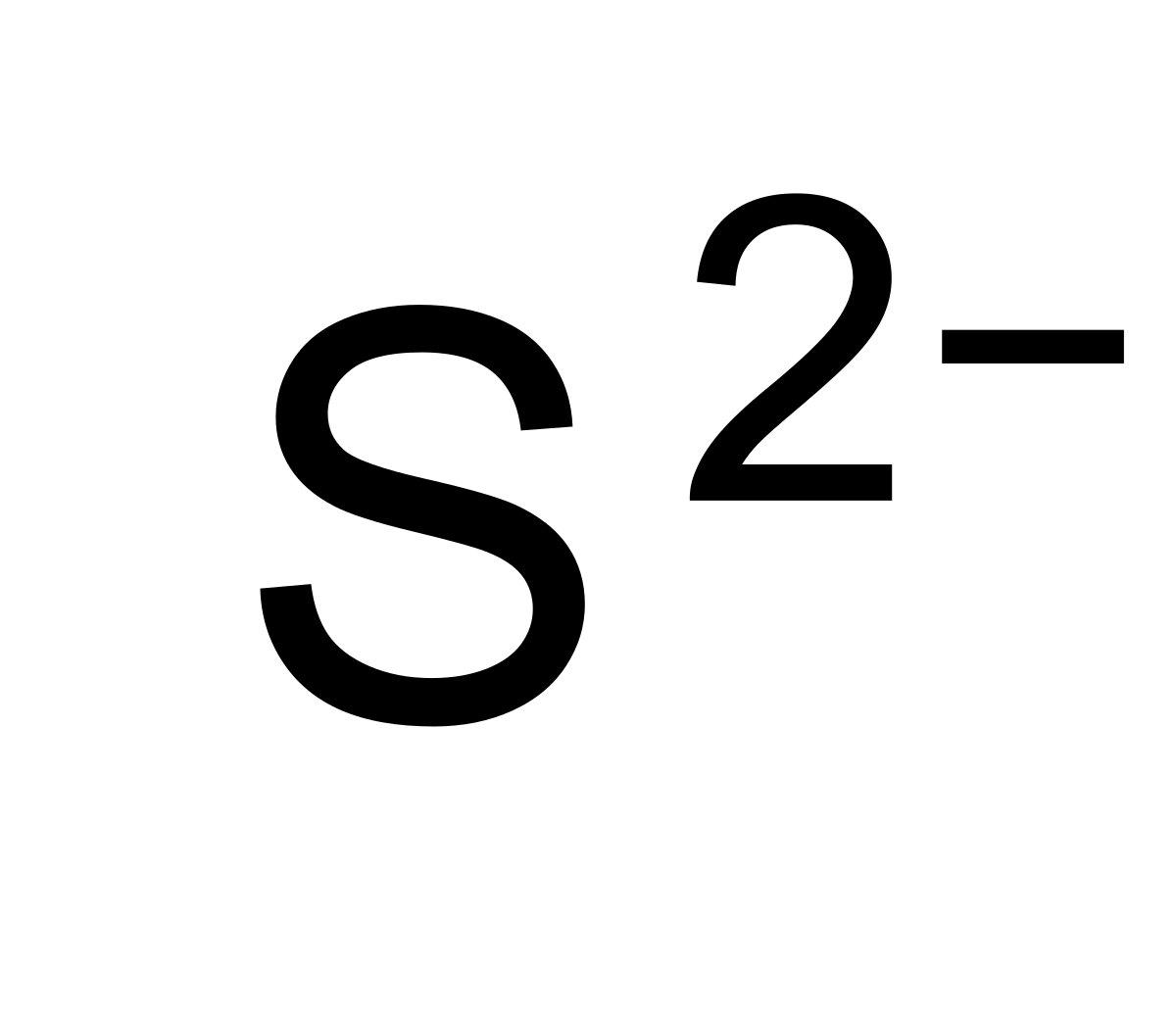What Is The Charge Of Sulfide
What Is The Charge Of Sulfide - For example, iron( ii ) has a 2+ charge; Roman numeral notation indicates charge of ion when element commonly forms more than one ion. In your case, the sulfide anion, s2−, carries a (2 −) negative charge, which can only mean that it gained electrons.
For example, iron( ii ) has a 2+ charge; Roman numeral notation indicates charge of ion when element commonly forms more than one ion. In your case, the sulfide anion, s2−, carries a (2 −) negative charge, which can only mean that it gained electrons.
For example, iron( ii ) has a 2+ charge; In your case, the sulfide anion, s2−, carries a (2 −) negative charge, which can only mean that it gained electrons. Roman numeral notation indicates charge of ion when element commonly forms more than one ion.
Schematic illustration of the different types of interphases between
For example, iron( ii ) has a 2+ charge; Roman numeral notation indicates charge of ion when element commonly forms more than one ion. In your case, the sulfide anion, s2−, carries a (2 −) negative charge, which can only mean that it gained electrons.
Sulfide charge atilagrid
In your case, the sulfide anion, s2−, carries a (2 −) negative charge, which can only mean that it gained electrons. For example, iron( ii ) has a 2+ charge; Roman numeral notation indicates charge of ion when element commonly forms more than one ion.
Sulfide Wikipedia
For example, iron( ii ) has a 2+ charge; Roman numeral notation indicates charge of ion when element commonly forms more than one ion. In your case, the sulfide anion, s2−, carries a (2 −) negative charge, which can only mean that it gained electrons.
We can draw three inequivalent Lewis structures for carbonyl sulfide
In your case, the sulfide anion, s2−, carries a (2 −) negative charge, which can only mean that it gained electrons. For example, iron( ii ) has a 2+ charge; Roman numeral notation indicates charge of ion when element commonly forms more than one ion.
How to Write the Formula for Sulfide ion YouTube
In your case, the sulfide anion, s2−, carries a (2 −) negative charge, which can only mean that it gained electrons. Roman numeral notation indicates charge of ion when element commonly forms more than one ion. For example, iron( ii ) has a 2+ charge;
Sulfate définition illustrée et explications
Roman numeral notation indicates charge of ion when element commonly forms more than one ion. For example, iron( ii ) has a 2+ charge; In your case, the sulfide anion, s2−, carries a (2 −) negative charge, which can only mean that it gained electrons.
SOLVEDFor each of the following ions, write the full electron
Roman numeral notation indicates charge of ion when element commonly forms more than one ion. For example, iron( ii ) has a 2+ charge; In your case, the sulfide anion, s2−, carries a (2 −) negative charge, which can only mean that it gained electrons.
Chargedischarge profiles of bismuth sulfide recorded at 2 A g −1 (a
In your case, the sulfide anion, s2−, carries a (2 −) negative charge, which can only mean that it gained electrons. Roman numeral notation indicates charge of ion when element commonly forms more than one ion. For example, iron( ii ) has a 2+ charge;
Sulfide charge jordoutdoor
Roman numeral notation indicates charge of ion when element commonly forms more than one ion. For example, iron( ii ) has a 2+ charge; In your case, the sulfide anion, s2−, carries a (2 −) negative charge, which can only mean that it gained electrons.
Insights into interfacial physiochemistry in sulfide solidstate
In your case, the sulfide anion, s2−, carries a (2 −) negative charge, which can only mean that it gained electrons. For example, iron( ii ) has a 2+ charge; Roman numeral notation indicates charge of ion when element commonly forms more than one ion.
Roman Numeral Notation Indicates Charge Of Ion When Element Commonly Forms More Than One Ion.
For example, iron( ii ) has a 2+ charge; In your case, the sulfide anion, s2−, carries a (2 −) negative charge, which can only mean that it gained electrons.