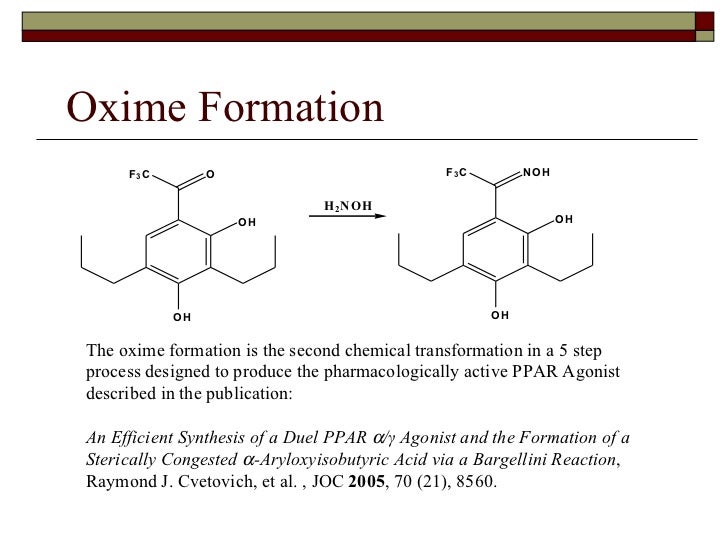
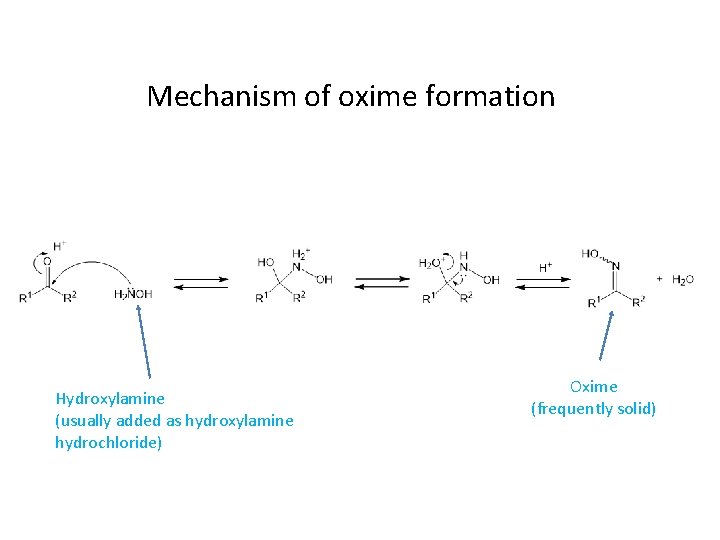
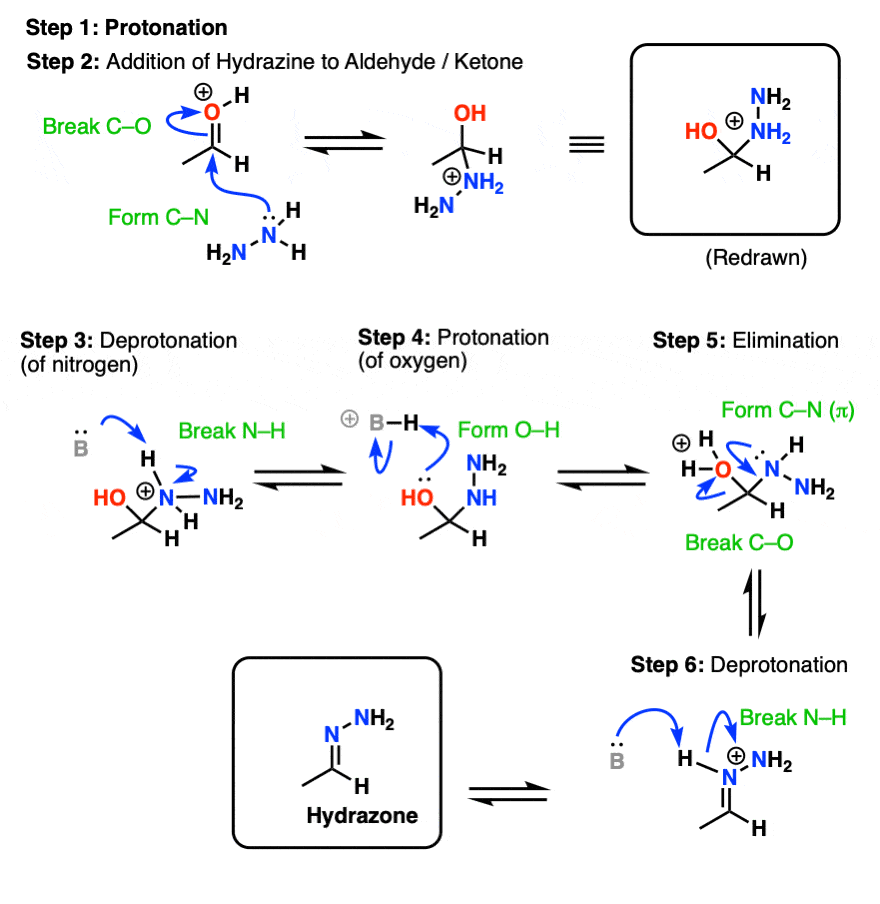
Oxime Formation Mechanism
Oxime Formation Mechanism - Reaction of aldehydes and ketones with hydroxylamine gives oximes. The nucleophilicity of the nitrogen on the hydroxylamine is. The first step in the process is formation of an oxime from the aldehyde or ketone, which occurs in a sequence similar to.
The first step in the process is formation of an oxime from the aldehyde or ketone, which occurs in a sequence similar to. Reaction of aldehydes and ketones with hydroxylamine gives oximes. The nucleophilicity of the nitrogen on the hydroxylamine is.
Reaction of aldehydes and ketones with hydroxylamine gives oximes. The nucleophilicity of the nitrogen on the hydroxylamine is. The first step in the process is formation of an oxime from the aldehyde or ketone, which occurs in a sequence similar to.
Nitrile Oxide Synthesis Via Oxime
The first step in the process is formation of an oxime from the aldehyde or ketone, which occurs in a sequence similar to. Reaction of aldehydes and ketones with hydroxylamine gives oximes. The nucleophilicity of the nitrogen on the hydroxylamine is.
[PDF] Hydrolytic stability of hydrazones and oximes. Semantic Scholar
Reaction of aldehydes and ketones with hydroxylamine gives oximes. The first step in the process is formation of an oxime from the aldehyde or ketone, which occurs in a sequence similar to. The nucleophilicity of the nitrogen on the hydroxylamine is.
Formation of oximes and hydrazones Aldehydes and ketones Organic
The nucleophilicity of the nitrogen on the hydroxylamine is. Reaction of aldehydes and ketones with hydroxylamine gives oximes. The first step in the process is formation of an oxime from the aldehyde or ketone, which occurs in a sequence similar to.
Oxime Formation Step Optimization
The nucleophilicity of the nitrogen on the hydroxylamine is. The first step in the process is formation of an oxime from the aldehyde or ketone, which occurs in a sequence similar to. Reaction of aldehydes and ketones with hydroxylamine gives oximes.
cyclohexanone oxime synthesis mechanism chemicalreaction YouTube
The first step in the process is formation of an oxime from the aldehyde or ketone, which occurs in a sequence similar to. Reaction of aldehydes and ketones with hydroxylamine gives oximes. The nucleophilicity of the nitrogen on the hydroxylamine is.
Proposed mechanistic pathway for the formation of the oximeoxime
The nucleophilicity of the nitrogen on the hydroxylamine is. The first step in the process is formation of an oxime from the aldehyde or ketone, which occurs in a sequence similar to. Reaction of aldehydes and ketones with hydroxylamine gives oximes.
[Solved] Q2. I need the oxime formation mechanism and the Beckmann
The nucleophilicity of the nitrogen on the hydroxylamine is. The first step in the process is formation of an oxime from the aldehyde or ketone, which occurs in a sequence similar to. Reaction of aldehydes and ketones with hydroxylamine gives oximes.
Oxime
The nucleophilicity of the nitrogen on the hydroxylamine is. The first step in the process is formation of an oxime from the aldehyde or ketone, which occurs in a sequence similar to. Reaction of aldehydes and ketones with hydroxylamine gives oximes.
Other Reactions of Ketones and Aldehydes Relative Reactivity
Reaction of aldehydes and ketones with hydroxylamine gives oximes. The first step in the process is formation of an oxime from the aldehyde or ketone, which occurs in a sequence similar to. The nucleophilicity of the nitrogen on the hydroxylamine is.
Imines Properties, Formation, Reactions, and Mechanisms Master
Reaction of aldehydes and ketones with hydroxylamine gives oximes. The first step in the process is formation of an oxime from the aldehyde or ketone, which occurs in a sequence similar to. The nucleophilicity of the nitrogen on the hydroxylamine is.
Reaction Of Aldehydes And Ketones With Hydroxylamine Gives Oximes.
The first step in the process is formation of an oxime from the aldehyde or ketone, which occurs in a sequence similar to. The nucleophilicity of the nitrogen on the hydroxylamine is.
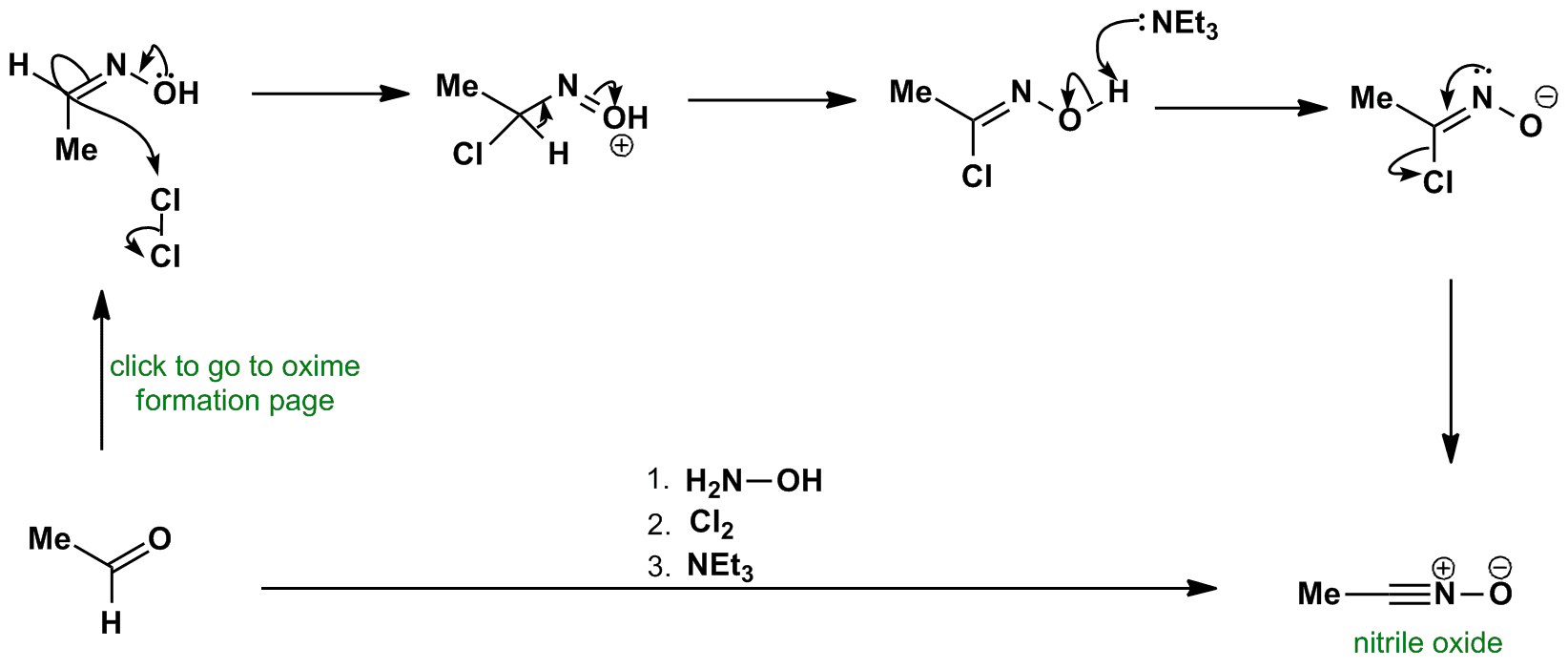
![[PDF] Hydrolytic stability of hydrazones and oximes. Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/5dd85edc0941ee0d75a381fc4002f12644b2bc2f/10-Figure5-1.png)